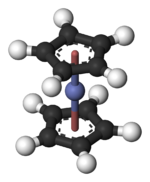
Cobaltocene
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Cobaltocene
Bis(η5-cyclopentadienyl)cobalt | |||
| Other names
Cp2Co
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.013.692 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| [Co(η5-C5H5)2] | |||
| Molar mass | 189.12 g/mol | ||
| Appearance | Dark purple solid | ||
| Melting point | 171–173 °C (340–343 °F; 444–446 K) | ||
| Insoluble | |||
| Structure | |||
| sandwich | |||
| zero | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
236 J K−1 mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
+237 kJ/mol (uncertain) | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−5839 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
  [1] [1]
| |||
| Danger[1] | |||
| H228, H317, H351[1] | |||
| P210, P261, P280, P363, P405, P501[1] | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External SDS | ||
| Related compounds | |||
Related metallocenes
|
Ferrocene Nickelocene Rhodocene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cobaltocene, known also as bis(cyclopentadienyl)cobalt(II) or even "bis Cp cobalt", is an organocobalt compound with the formula Co(C5H5)2. It is a dark purple solid that sublimes readily slightly above room temperature. Cobaltocene was discovered shortly after ferrocene, the first metallocene. Due to the ease with which it reacts with oxygen, the compound must be handled and stored using air-free techniques.
Synthesis
[edit]
Cobaltocene is prepared by the reaction of sodium cyclopentadienide (NaC5H5) with anhydrous cobalt(II) chloride in THF solution. Sodium chloride is cogenerated, and the organometallic product is usually purified by vacuum sublimation.[2]
Structure and bonding
[edit]In Co(C5H5)2 the Co centre is "sandwiched" between two cyclopentadienyl (Cp) rings. The Co–C bond lengths are about 2.1 Å, slightly longer than the Fe–C bond in ferrocene.[3]
Co(C5H5)2 belongs to a group of organometallic compounds called metallocenes or sandwich compounds.[4] Cobaltocene has 19 valence electrons, one more than usually found in organotransition metal complexes such as its very stable relative ferrocene. (See 18-electron rule.) This additional electron occupies an orbital that is antibonding with respect to the Co–C bonds. Consequently, the Co–C distances are slightly longer than the Fe–C bonds in ferrocene. Many chemical reactions of Co(C5H5)2 are characterized by its tendency to lose this "extra" electron, yielding an 18-electron cation known as cobaltocenium:
The otherwise close relative of cobaltocene, rhodocene does not exist as a monomer, but spontaneously dimerizes by formation of a C–C bond between Cp rings.
Reactions
[edit]Redox properties
[edit]Co(C5H5)2 is a common one-electron reducing agent in the laboratory.[5] In fact, the reversibility of the Co(C5H5)2 redox couple is so well-behaved that Co(C5H5)2 may be used in cyclic voltammetry as an internal standard. Its permethylated analogue decamethylcobaltocene (Co(C5Me5)2) is an especially powerful reducing agent, due to inductive donation of electron density from the 10 methyl groups, prompting the cobalt to give up its "extra" electron even more so. These two compounds are rare examples of reductants that dissolve in non-polar organic solvents. The reduction potentials of these compounds follow, using the ferrocene-ferrocenium couple as the reference:
| Half-reaction | E0 (V) |
|---|---|
| Fe(C 5H 5)+ 2 + e− ⇌ Fe(C5H5)2 |
0.00 (by definition) |
| Fe(C 5Me 5)+ 2 + e− ⇌ Fe(C5Me5)2 |
−0.59 |
| Co(C 5H 5)+ 2 + e− ⇌ Co(C5H5)2 |
−1.33 |
| Co(C 5Me 5)+ 2 + e− ⇌ Co(C5Me5)2 |
−1.94 |
The data show that the decamethyl compounds are around 600 mV more reducing than the parent metallocenes. This substituent effect is, however, overshadowed by the influence of the metal: changing from Fe to Co renders the reduction more favorable by over 1.3 volts.
Carbonylation
[edit]Treatment of Co(C5H5)2 with carbon monoxide gives the cobalt(I) derivative Co(C5H5)(CO)2, concomitant with loss of one Cp ligand. This conversion is conducted near 130 °C with 500 psi of CO.[2][6]
See also
[edit]References
[edit]- ^ a b c d "Bis(cyclopentadienyl)cobalt(II)". American Elements. Retrieved 2018-08-24.
- ^ a b King, R. B. (1965). Organometallic Syntheses. Vol. 1. New York, NY: Academic Press.
- ^ Antipin, M. Yu.; Boese, R.; Augart, N.; Schmid, G. (1993). "Redetermination of the cobaltocene crystal structure at 100 K and 297 K: Comparison with ferrocene and nickelocene". Struct. Chem. 4 (2): 91–101. doi:10.1007/BF00677370. S2CID 93871667.
- ^ Elschenbroich, C.; Salzer, A. (1992). Organometallics: A Concise Introduction (2nd ed.). Weinheim: Wiley-VCH. ISBN 978-3-527-28165-7.
- ^ Connelly, N. G.; Geiger, W. E. (1996). "Chemical Redox Agents for Organometallic Chemistry". Chem. Rev. 96 (2): 877–910. doi:10.1021/cr940053x. PMID 11848774.
- ^ King, R. B.; Stone, F. G. A. (1967). Cyclopentadienyl Metal Carbonyls and Some Derivatives. Inorganic Syntheses. Vol. 7. pp. 99–115. doi:10.1002/9780470132388.ch31. ISBN 9780470132388.